UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14D-9
Solicitation/Recommendation Statement
Under Section 14(d)(4) of the Securities Exchange Act of 1934
TOURMALINE BIO, INC.
(Name of Subject Company)
TOURMALINE BIO, INC.
(Name of Person(s) Filing Statement)
Common Stock, $0.0001 par value per share
(Title of Class of Securities)
89157D105
(CUSIP Number of Class of Securities)
Sandeep Kulkarni
Chief Executive Officer
Tourmaline Bio, Inc.
27 West 24th Street, Suite 702
New York, NY 10010
(646) 481-9832
(Name, Address and Telephone Number of Person Authorized to Receive Notices and Communications
on Behalf of the Person Filing Statement)
With copies to:
William Sorabella, Esq.
Brandon Fenn, Esq.
Cooley LLP
55 Hudson Yards
New York, NY 10001
(212) 479-6000
| ☒ | Check the box if the filing relates solely to preliminary communications made before the commencement of a tender offer. |
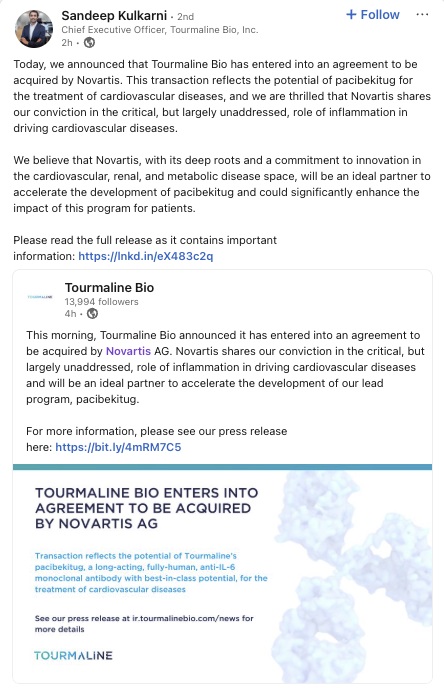
This Schedule 14D-9 filing consists of certain communications relating to the proposed acquisition of Tourmaline Bio, Inc., a Delaware corporation (the “Company” or “Tourmaline”), by Novartis AG, a company limited by shares (Aktiengesellschaft) incorporated under the laws of Switzerland (“Parent” or “Novartis”), pursuant to the terms and subject to the conditions of an Agreement and Plan of Merger, dated as of September 8, 2025 (the “Merger Agreement”), by and among the Company, Parent and Torino Merger Sub Inc., a Delaware corporation and an indirect wholly owned subsidiary of Parent (“Purchaser”). Pursuant to the Merger Agreement, upon the terms and subject to the conditions thereof, Purchaser will commence a cash tender offer (the “Offer”) no later than September 29, 2025. The Offer will consist of an offer to purchase all of the outstanding shares of common stock of the Company, par value $0.0001 per share (the “Shares”), at a price of $48.00 per Share, in cash, without interest, and subject to any withholding of taxes required by applicable legal requirements (the “Offer Price”). Following the date and time of the irrevocable acceptance for payment by Purchaser of the Shares that have been validly tendered and not validly withdrawn pursuant to and subject to the conditions of the Offer (the “Offer Acceptance Time”) and subject to the satisfaction or waiver of certain conditions set forth in the Merger Agreement, Parent, Purchaser and the Company will, pursuant to Section 251(h) of the General Corporation Law of the State of Delaware (the “DGCL”) without a vote of the Company stockholders, effect a merger of Purchaser with and into the Company (the “Merger” and, together with the Offer and the other transactions contemplated by the Merger Agreement, the “Transactions”), with the Company continuing as the surviving corporation of the Merger and an indirect wholly owned subsidiary of Parent.
This Schedule 14D-9 filing consists of the following documents relating to the proposed Offer and the Merger:
| 1. | Exhibit 99.1: Postings by the Company and Sandeep Kulkarni, M.D., Chief Executive Officer of the Company, via LinkedIn, X and Bluesky on September 9, 2025; and |
| 2. | Exhibit 99.2: Email from Novartis to the Company’s employees, first used September 9, 2025. |
Forward-Looking Statements
This communication contains forward-looking statements that involve risks and uncertainties relating to future events and the future performance of Tourmaline and Novartis, including statements relating to the ability to complete and the timing of completion of the Transactions contemplated by the Merger Agreement, including the anticipated occurrence, manner and timing of the proposed Offer, the parties’ ability to satisfy the conditions to the consummation of the Offer and the other conditions to the consummation of the Merger set forth in the Merger Agreement, the possibility of any termination of the Merger Agreement, and the prospective benefits of the proposed Transactions, and other statements that are not historical facts. The forward-looking statements contained in this communication are based on current expectations and assumptions that are subject to risks and uncertainties which may cause actual results to differ materially from the forward-looking statements. These statements may contain words such as “may,” “will,” “would,” “could,” “expect,” “anticipate,” “intend,” “plan,” “believe,” “estimate,” “project,” “seek,” “should,” “strategy,” “future,” “opportunity,” “potential” or other similar words and expressions indicating future results. Risks that may cause these forward-looking statements to be inaccurate include, without limitation: uncertainties as to the timing of the Offer; uncertainties as to how many of Tourmaline’s stockholders will tender their stock in the Offer; the possibility that competing offers or acquisition proposals will be made; the possibility that various closing conditions for the Transactions may not be satisfied or waived, including that a governmental entity may prohibit, delay, or refuse to grant approval for the consummation of the Transactions (or only grant approval subject to adverse conditions or limitations); the difficulty of predicting the timing or outcome of regulatory approvals or actions, if any; the occurrence of any event, change or other circumstance that could give rise to the termination of the Merger Agreement; the possibility that the Transactions do not close; risks related to the parties’ ability to realize the anticipated benefits of the proposed Transactions, including the possibility that the expected benefits from the proposed acquisition will not be realized or will not be realized within the expected time period and that Tourmaline and Novartis will not be integrated successfully or that such integration may be more difficult, time-consuming or costly than expected; the effects of the Transactions on relationships with employees, suppliers, manufacturers, other business partners or governmental entities; negative effects of this announcement or the consummation of the proposed Transactions on the market price of Tourmaline’s common stock and/or Tourmaline’s operating results; significant transaction costs; unknown or inestimable liabilities; the risk of litigation and/or regulatory actions related to the proposed Transactions; Novartis’s ability to fund the proposed Transactions; the time-consuming and uncertain regulatory approval process; the uncertainties inherent in the costly and time-consuming therapeutic product development process and the uncertainty of clinical success, including risks related to failure or delays in successfully
1
initiating or completing clinical trials and assessing patients; global economic, financial, and healthcare system disruptions and the current and potential future negative impacts to the parties’ business operations and financial results; the sufficiency of the parties’ cash flows and capital resources; the parties’ ability to achieve targeted or expected future financial performance and results and the uncertainty of future tax, accounting and other provisions and estimates; and other risks and uncertainties affecting Tourmaline, including those described from time to time under the caption “Risk Factors” and elsewhere in Tourmaline’s filings and reports with the U.S. Securities and Exchange Commission (the “SEC”), including Tourmaline’s Annual Report on Form 10-K for the fiscal year ended December 31, 2024 and Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2025, as well as the Tender Offer Statement on Schedule TO and related tender offer documents to be filed by Novartis and the Purchaser, and the Solicitation/Recommendation Statement on Schedule 14D-9 to be filed by Tourmaline. Any forward-looking statements are made based on the current beliefs and judgments of Tourmaline’s and Novartis’s management, and the reader is cautioned not to rely on any forward-looking statements made by Tourmaline or Novartis. Except as required by law, Tourmaline and Novartis do not undertake any obligation to update (publicly or otherwise) any forward-looking statement, including without limitation any financial projection or guidance, whether as a result of new information, future events, or otherwise.
Important Information about the Tender Offer and Where to Find It
The Offer for Tourmaline outstanding common stock referred to in this communication has not yet commenced. The description contained in this communication is neither a recommendation, nor an offer to purchase nor a solicitation of an offer to sell any shares of common stock of Tourmaline or any other securities, nor is it a substitute for the tender offer materials that Novartis and the Purchaser will file with the SEC. The solicitation and offer to purchase Tourmaline’s common stock will only be made pursuant to an offer to purchase and related tender offer materials. At the time the Offer is commenced, Novartis and the Purchaser will file a tender offer statement on Schedule TO and thereafter Tourmaline will file a solicitation/recommendation statement on Schedule 14D-9 with the SEC with respect to the Offer.
THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND CERTAIN OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT ON SCHEDULE 14D-9, AS THEY MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, WILL CONTAIN IMPORTANT INFORMATION, INCLUDING THE TERMS AND CONDITIONS OF THE OFFER. ANY HOLDERS OF TOURMALINE SHARES ARE URGED TO READ THESE DOCUMENTS CAREFULLY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION THAT HOLDERS SHOULD CONSIDER BEFORE MAKING ANY DECISION REGARDING TENDERING THEIR SHARES.
The offer to purchase, the letter of transmittal, the solicitation/recommendation statement and related offer documents will be made available for free at the SEC’s website at www.sec.gov. Copies of those offer documents and all other documents filed by Novartis, the Purchaser and Tourmaline will be made available at no charge by directing a request to the information agent for the Offer, which will be named in the Schedule TO to be filed with the SEC. Copies of the solicitation/recommendation statement on Schedule 14D-9 to be filed with the SEC by Tourmaline will be available free of charge on Tourmaline’s investor relations website at https://ir.tourmalinebio.com/.
In addition, Tourmaline files annual, quarterly and current reports and other information with the SEC, which are also made available free of charge on the Company’s investor relations website at https://ir.tourmalinebio.com/ and at the SEC’s website at www.sec.gov.
2
Exhibit 99.1
Tourmaline LinkedIn Post
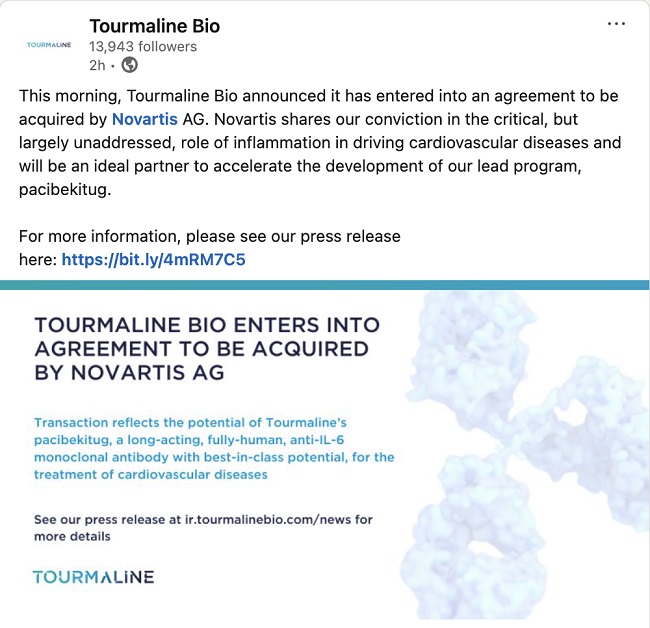
Sandeep Kulkarni, M.D. LinkedIn Post
Tourmaline X Post

Tourmaline Bluesky Post

Forward-Looking Statements
This communication contains forward-looking statements that involve risks and uncertainties relating to future events and the future performance of Tourmaline and Novartis, including statements relating to the ability to complete and the timing of completion of the Transactions contemplated by the Merger Agreement, including the anticipated occurrence, manner and timing of the proposed Offer, the parties’ ability to satisfy the conditions to the consummation of the Offer and the other conditions to the consummation of the Merger set forth in the Merger Agreement, the possibility of any termination of the Merger Agreement, and the prospective benefits of the proposed Transactions, and other statements that are not historical facts. The forward-looking statements contained in this communication are based on current expectations and assumptions that are subject to risks and uncertainties which may cause actual results to differ materially from the forward-looking statements. These statements may contain words such as “may,” “will,” “would,” “could,” “expect,” “anticipate,” “intend,” “plan,” “believe,” “estimate,” “project,” “seek,” “should,” “strategy,” “future,” “opportunity,” “potential” or other similar words and expressions indicating future results. Risks that may cause these forward-looking statements to be inaccurate include, without limitation: uncertainties as to the timing of the Offer; uncertainties as to how many of Tourmaline’s stockholders will tender their stock in the Offer; the possibility that competing offers or acquisition proposals will be made; the possibility that various closing conditions for the Transactions may not be satisfied or waived, including that a governmental entity may prohibit, delay, or refuse to grant approval for the consummation of the Transactions (or only grant approval subject to adverse conditions or limitations); the difficulty of predicting the timing or outcome of regulatory approvals or actions, if any; the occurrence of any event, change or other circumstance that could give rise to the termination of the Merger Agreement; the possibility that the Transactions do not close; risks related to the parties’ ability to realize the anticipated benefits of the proposed Transactions, including the possibility that the expected benefits from the proposed acquisition will not be realized or will not be realized within the expected time period and that Tourmaline and Novartis will not be integrated successfully or that such integration may be more difficult, time-consuming or costly than expected; the effects of the Transactions on relationships with employees, suppliers, manufacturers, other business partners or governmental entities; negative effects of this announcement or the consummation of the proposed Transactions on the market price of Tourmaline’s common stock and/or Tourmaline’s operating results; significant transaction costs; unknown or inestimable liabilities; the risk of litigation and/or regulatory actions related to the proposed Transactions; Novartis’s ability to fund the proposed Transactions; the time-consuming and uncertain regulatory approval process; the uncertainties inherent in the costly and time-consuming therapeutic product development process and the uncertainty of clinical success, including risks related to failure or delays in successfully initiating or completing clinical trials and assessing patients; global economic, financial, and healthcare system disruptions and the current and potential future negative impacts to the parties’ business operations and financial results; the sufficiency of the parties’ cash flows and capital resources; the parties’ ability to achieve targeted or expected future financial performance and results and the uncertainty of future tax, accounting and other provisions and estimates; and other risks and uncertainties affecting Tourmaline, including those described from time to time under the caption “Risk Factors” and elsewhere in Tourmaline’s filings and reports with the U.S. Securities and Exchange Commission (the “SEC”), including Tourmaline’s Annual Report on Form 10-K for the fiscal year ended December 31, 2024 and Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2025, as well as the Tender Offer Statement on Schedule TO and related tender offer documents to be filed by Novartis and the Purchaser, and the Solicitation/Recommendation Statement on Schedule 14D-9 to be filed by Tourmaline. Any forward-looking statements are made based on the current beliefs and judgments of Tourmaline’s and Novartis’s management, and the reader is cautioned not to rely on any forward-looking statements made by Tourmaline or Novartis. Except as required by law, Tourmaline and Novartis do not undertake any obligation to update (publicly or otherwise) any forward-looking statement, including without limitation any financial projection or guidance, whether as a result of new information, future events, or otherwise.
Important Information about the Tender Offer and Where to Find It
The Offer for Tourmaline outstanding common stock referred to in this communication has not yet commenced. The description contained in this communication is neither a recommendation, nor an offer to purchase nor a solicitation of an offer to sell any shares of common stock of Tourmaline or any other securities, nor is it a substitute for the tender offer materials that Novartis and the Purchaser will file with the SEC. The solicitation and offer to purchase Tourmaline’s common stock will only be made pursuant to an offer to purchase and related tender offer materials. At the time the Offer is commenced, Novartis and the Purchaser will file a tender offer statement on Schedule TO and thereafter Tourmaline will file a solicitation/recommendation statement on Schedule 14D-9 with the SEC with respect to the Offer.
THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND CERTAIN OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT ON SCHEDULE 14D-9, AS THEY MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, WILL CONTAIN IMPORTANT INFORMATION, INCLUDING THE TERMS AND CONDITIONS OF THE OFFER. ANY HOLDERS OF TOURMALINE SHARES ARE URGED TO READ THESE DOCUMENTS CAREFULLY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION THAT HOLDERS SHOULD CONSIDER BEFORE MAKING ANY DECISION REGARDING TENDERING THEIR SHARES.
The offer to purchase, the letter of transmittal, the solicitation/recommendation statement and related offer documents will be made available for free at the SEC’s website at www.sec.gov. Copies of those offer documents and all other documents filed by Novartis, the Purchaser and Tourmaline will be made available at no charge by directing a request to the information agent for the Offer, which will be named in the Schedule TO to be filed with the SEC. Copies of the solicitation/recommendation statement on Schedule 14D-9 to be filed with the SEC by Tourmaline will be available free of charge on Tourmaline’s investor relations website at https://ir.tourmalinebio.com/.
In addition, Tourmaline files annual, quarterly and current reports and other information with the SEC, which are also made available free of charge on the Company’s investor relations website at https://ir.tourmalinebio.com/ and at the SEC’s website at www.sec.gov.
Exhibit 99.2
FROM: Sandeep Kulkarni
TO: tourmaline-allhands
SUBJECT: Welcome message from Dr. Ruchira Glaser
Dear Tourmaline Team,
As a follow up to the discussion from earlier today, I’m pleased to share a welcome message from Dr. Ruchira Glaser, M.D., M.S., Development Unit Head, Cardiovascular, Renal and Metabolic at Novartis. Please see the message attached.
Sandeep

|

|
Welcoming Tourmaline Bio into the Novartis family
Dear Tourmaline Team,
On behalf of my Novartis colleagues, I want to express our genuine excitement to partner with you to advance the development of innovative therapies for patients living with cardiovascular diseases. From our initial interactions with your leadership to our deeper collaborative discussions, we’ve been thoroughly impressed by the people, expertise, and capabilities you’ve built. Most importantly, we are inspired by your dedication and passion for innovation, which closely aligns with our own values and culture.
At Novartis, our focus is on delivering high-value medicines that alleviate society’s greatest health challenges. Pacibekitug is an important addition to our cardiovascular pipeline, addressing residual inflammatory risk in ASCVD—a critical unmet need. We are committed to progressing this promising asset, leveraging our combined expertise to bring transformative solutions to patients in need.
Your dedication and hard work have been instrumental in successfully bringing pacibekitug to its current stage of development. At Novartis, we are dedicated to fostering a culture that brings out the best in everyone, enabling us to continue reimagining medicine for a better and longer life for patients. Working collaboratively on groundbreaking scientific and medical advancements makes both of our companies exceptional places to work. Upon closing of the transaction, estimated for the fourth quarter 2025 and subject to customary conditions, we eagerly anticipate learning from you, supporting you, and joining forces to advance cardiovascular health and bring medical innovation to patients globally.
I would like to extend my gratitude to the Tourmaline leadership team and colleagues who have been pivotal in forging this partnership. In the coming weeks, post-transaction closure, you will learn more about our collaborative efforts, but until then, we will continue to operate as separate entities.
We look forward to connecting with you after the transaction closes to redefine cardiovascular treatment and make a meaningful impact on patients’ lives worldwide.
Thank you.
Best regards,
Ruchira Glaser, M.D., M.S.
Development Unit Head, Cardiovascular, Renal and Metabolic
Novartis
External Communications
Business Use Only

|

|
……………………………….
Important Information about the Tender Offer
This communication is neither an offer to purchase nor a solicitation of an offer to sell any shares of the common stock, par value USD 0.0001 (the “Shares”), of Tourmaline or any other securities. The tender offer for the outstanding Shares described in this communication has not commenced. At the time the tender offer is commenced, Novartis and its indirect wholly owned subsidiary, Torino Merger Sub Inc. (“Purchaser”), will file a tender offer statement on Schedule TO, including an offer to purchase, a letter of transmittal and related documents, with the U.S. Securities and Exchange Commission (the “SEC”), and Tourmaline will file a solicitation/recommendation statement on Schedule 14D-9 with the SEC, in each case with respect to the tender offer.
INVESTORS AND SECURITY HOLDERS ARE URGED TO CAREFULLY READ BOTH THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A LETTER OF TRANSMITTAL AND RELATED DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT ON SCHEDULE 14D-9 REGARDING THE TENDER OFFER, AS THEY MAY BE AMENDED FROM TIME TO TIME, WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION THAT INVESTORS AND SECURITY HOLDERS SHOULD CONSIDER BEFORE MAKING ANY DECISION REGARDING TENDERING THEIR SECURITIES.
An offer to purchase the Shares will only be made pursuant to the offer to purchase, the letter of transmittal and related offer documents filed as a part of the Schedule TO. Those materials and all other documents filed by, or caused to be filed by, Novartis, Purchaser and Tourmaline with the SEC will be available at no charge on the SEC’s website at www.sec.gov or by directing such requests to the information agent for the offer, which will be named in the tender offer statement. The offer to purchase and related materials also may be obtained for free under the “Investors – Financial Data” section of Novartis AG’s website at www.novartis.com/investors/financial-data/sec-filings. The solicitation/recommendation statement also may be obtained for free under the “Investors” section of Tourmaline’s website at ir.tourmalinebio.com. In addition, Tourmaline files annual, quarterly and current reports and other information, and Novartis files annual reports and other information with the SEC, which are also available to the public at no charge at www.sec.gov.
Forward-Looking Statements
This communication contains statements that are not statements of historical fact, or “forward-looking statements,” including with respect to Novartis’ proposed acquisition of Tourmaline. Forward-looking statements can generally be identified by words such as “potential,” “can,” “will,” “plan,” “may,” “could,” “would,” “expect,” “anticipate,” “look forward,” “believe,” “committed,” “investigational,” “pipeline,” “launch,” or similar terms, or by express or implied discussions regarding [potential marketing approvals, new indications or labeling for Tourmaline’s product candidates, Tourmaline’s platform, the proposed acquisition of
External Communications
Business Use Only

|

|
Tourmaline and the expected timetable for completing the proposed acquisition, the benefits sought to be achieved in the proposed acquisition, or potential future revenues from Tourmaline’s product candidates. You should not place undue reliance on these statements. Such forward-looking statements are based on Novartis’ current beliefs and expectations regarding future events and are subject to significant known and unknown risks and uncertainties. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those set forth in the forward-looking statements. There can be no guarantee that clinical trials for any of Tourmaline’s product candidates will be successful, that Tourmaline’s approach to the discovery and development of product candidates based on its AOC™ platform will produce any products of commercial value, that any of Tourmaline’s product candidates will be submitted for marketing approval or approved for sale or, if approved, receive approval for any additional indications or labeling, in any market, or at any particular time, nor can there be any guarantee that, if approved, any of Tourmaline’s product candidates will be commercially successful in the future. Neither can there be any guarantee that the conditions to the closing of the proposed acquisition will be satisfied on the expected timetable or at all or that the expected benefits or synergies from this transaction will be achieved in the expected timeframe, or at all. In particular, expectations regarding Tourmaline or the transaction described in this communication could be affected by, among other things, the timing of the offer and the satisfaction of customary closing conditions, including the tender of a majority of the outstanding shares of Tourmaline common stock and the receipt of regulatory approvals on acceptable terms or at all; the risk that competing offers or acquisition proposals will be made; the effects of disruption from the transactions contemplated by the merger agreement and the impact of the announcement and pendency of the transactions on Novartis and/or Tourmaline’s businesses, including their relationships with employees, business partners or governmental entities; the risk that the offer or the merger may be more expensive to complete than anticipated; the risk that stockholder litigation in connection with the offer or the merger may result in significant costs of defense, indemnification and liability; a diversion of management’s attention from ongoing business operations and opportunities as a result of the offer, the merger or otherwise; general industry conditions and competition; general political, economic and business conditions, including interest rate and currency exchange rate fluctuations; the uncertainties inherent in research and development, including clinical trial results and additional analysis of existing clinical data; regulatory actions or delays or government regulation generally; global trends toward health care cost containment, including government, payor and general public pricing and reimbursement pressures and requirements for increased pricing transparency; our ability to obtain or maintain proprietary intellectual property protection; the particular prescribing preferences of physicians and patients; general political, economic and business conditions; safety, quality, data integrity or manufacturing issues; potential or actual data security and data privacy breaches, or disruptions of our information technology systems, and other risks and factors referred to in Novartis AG’s and Tourmaline’s filings and reports with the SEC, including Novartis AG’s Annual Report on Form 20-F for the year ended December 31, 2024, Tourmaline’s Annual Report on Form 10-K for the year ended December 31, 2024 and Quarterly Reports on Form 10-Q for the quarters ended March 31, 2025 and June 30, 2025, and any subsequent filings made by either party with the SEC, available on the SEC’s website at www.sec.gov. Novartis is providing the information in this communication as of this date and Novartis does not undertake any obligation to update any forward-looking statements contained in this communication as a result of new information, future events or otherwise, except to the extent required by law.
External Communications
Business Use Only
